 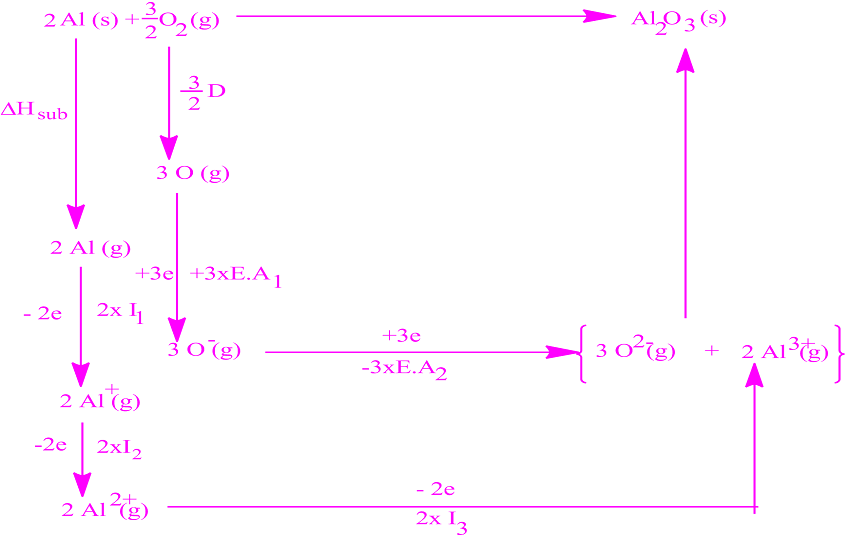
The distance between ions in the crystal are continously enlarged until the ions are very far apart.The energy required for this process is the lattice energy of NaCl.īorn- Haber cycle : The Born-Haber Cycle is Hess' Law applied to an ionic solid. Lattice energy is the change in energy that occurs when an ionic solid is seperated into isolated ions in the gas phase.For sodium
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
